
Facebook Twitter Instagram YouTube RSS Feed
Written on: March 11th, 2019 in Outreach
by Brittany Haywood, DNREC Wetland Monitoring and Assessment Program
 One part of our job is going out into the public and explaining simply our research and the benefits of wetlands. We often use the saying, “wetlands are like sponges” to describe their ability to absorb water, but recently we’ve been asked exactly how that is so.
One part of our job is going out into the public and explaining simply our research and the benefits of wetlands. We often use the saying, “wetlands are like sponges” to describe their ability to absorb water, but recently we’ve been asked exactly how that is so.
Describing wetlands as sponges dates back all the way to 1784 where Nicholas Turner described peat wetlands as, “in the nature of a sponge; for if a dry piece is put in water it will absorb double its weight” (Turner, 1757 p40). Since then this metaphor has been used all over the world to describe one of the many benefits that wetlands provide.
For this blog post we will explore the two linked concepts: one, how do sponges work, and two, how wetlands absorb water. Then, we will package it all up with a nice neat bow to make the connection that will hopefully satisfy all you knowledge seekers out there.
When you put a sponge in water, it absorbs it. This happens because the bubble like spaces inside the sponge have the ability to hold water and the sponge material locks it in. Water won’t be released until someone forces it out through squeezing or it could be allowed to dry out over time.
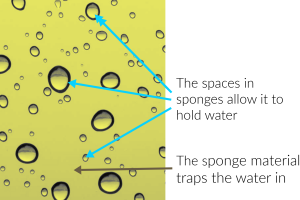 Also, have you ever noticed that it’s easier for a damp sponge to soak up water than a completely dry one? That’s because water is extremely sticky, and it sticks better to itself than anything else.
Also, have you ever noticed that it’s easier for a damp sponge to soak up water than a completely dry one? That’s because water is extremely sticky, and it sticks better to itself than anything else.
Nerdly speaking, water is a molecule made up of two hydrogen atoms and one oxygen atom. These molecules really like to form hydrogen bonds with each other, i.e. water loves to stick to itself. This is called cohesion. Water will also stick to other objects, like the dry sponge or soil, but it doesn’t do it quite as well. This is called adhesion. (Exploring Our Fluid Earth, 2019)
Wetlands are made up of wet soils, water loving plants, and of course, water. Soils have lots of spaces in between the soil particles called soil pores. And like the setup of the sponge, they can hold and trap water. How much water they hold onto depends on the pores sizes and soil type. Clay soils have small pores and trap water very well whereas sandy soils have larger pores allowing water to move more freely through it. Also these mineral type soils tend to attract water because of their unsatisfied ionic charge. (Nelson, 2015)
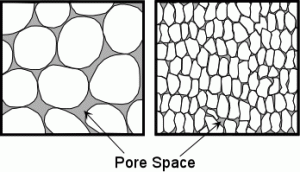
Pore spaces are in between the soil particles. The soil particles (soil material) trap the water. The two graphics represent two different soil types. (Nelson, 2015)
Wetland plants also play an important role in water absorption. These plants are specifically adapted to live in watery places. They absorb water and use what’s in it to grow. So not only do wetlands have water storage capabilities in their soils, they have it in their plants.
Water in wetlands is either released naturally and slowly over time to groundwater supplies, streams, or it evaporates. It can also be physically forced out by someone or something draining it.
As you have read above, water sticks to water better than dry objects so it would make sense that in order for a wetland to do its job, it needs to be wet. So draining wetlands may not be the best idea if you want to have a good functioning wetland.
So the long and short of it is both sponges and wetlands:

This wetland’s water carrying capacity has been reached and is now overflowing due to excess rainfall. (Lani Wall)
But wait, you’re probably thinking if wetlands absorb water, do they have a limit as to what they can hold?
Yes they do! Just like sponges, wetlands have a carrying capacity. So when the wetland becomes full you will see more pooling surface water, especially recently with all this rain we’ve been having in Delaware.
Now that you know how wetlands absorb and hold water, just think about how much more H2O you would see on our land surfaces if wetlands weren’t there or if a wetland’s soil pores got clogged up from super dirty water…
Exploring Our Fluid Earth. (n.d.). Hydrogen Bonds Make Water Sticky. Retrieved from https://manoa.hawaii.edu/exploringourfluidearth/chemical/properties-water/hydrogen-bonds-make-water-sticky
Nelson, S. A. (2015, November 11). Groundwater. Retrieved February 27, 2019, from https://www.tulane.edu/~sanelson/eens1110/groundwater.htm
Turner, Nicholas. An essay on draining and improving peat bogs; in which their nature and properties are fully considered. By Mr. Nicholas Turner, of Bignor, Sussex. London, MDCCLXXXIV. [1784]. Eighteenth Century Collections Online. Gale. CIC Penn State University. 25 Feb. 2019. <http://find.galegroup.com.ezaccess.libraries.psu.edu/ecco/infomark.do?&source=gale&prodId=ECCO&userGroupName=psucic&tabID=T001&docId=CW108495006&type=multipage&contentSet=ECCOArticles&version=1.0&docLevel=FASCIMILE>.